INTRODUCTION
Systemic lupus erythematosus (SLE) is a chronic multisystem inflammatory disease whose cause is unknown, and that affects the skin, kidneys, lungs, nervous system and particularly joints, so this disorder is of interest for all the radiological subspecialties. Musculoskeletal alterations are frequently found in SLE(1), and may be related to the disease activity or, many times, to the treatment adopted. Since 1982, the American College of Radiology considers that arthritis is one of the 11 criteria for classification of the disease(2). The degree of involvement may range from a transitory arthralgia to a severe presentation of deforming arthropathy, and it is estimated that at least 90% of patients will present some of such manifestations in the course of the disease(3). In spite of this high prevalence, little attention has been given to the theme, to the radiological presentations. Additionally, the many complications related to the disease, whether iatrogenic or not, are poorly recognized in the radiological community.
ARTHRALGIA, ARTHRITIS AND DEFORMING ARTHROPATHY
Arthralgia and arthritis are the most frequent articular manifestations, the latter classically described as non-erosive, migratory and reversible, involving principally wrists, knees, shoulders and hands (particularly proximal interphalangeal joints) in about 80% of lupus patients(4). An intrinsic characteristic of the SLE arthropathy is the possibility of association with deformities present in about 5% of patients(5), that still lacks well established etiopathogenic mechanisms, but that is believed to be related to some type of intrinsic capsuloligamentous and tendinous laxity in these patients(6). Bywaters has tried to define this form of deforming arthropathy in SLE, primarily based on the metacarpal axis deviation, whether reversible or not, and on the so-called Jaccouds index(7) (Table 1). Three different forms of deforming arthropathy in this disease were later proposed as follows: Jaccouds arthropathy (JA), rhupus hand and mild deforming arthropathy(8) (Figure 1).
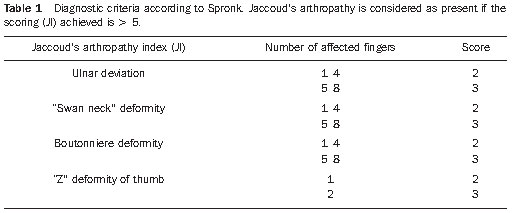
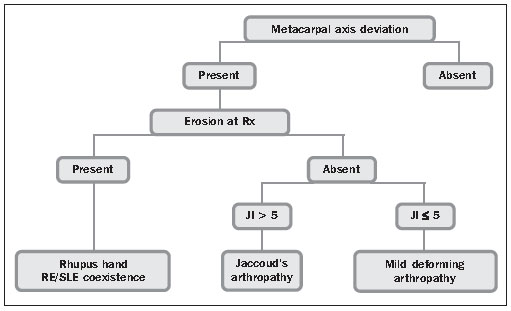
Figure 1. Algorithm with the forms of classification joint involvement in SLE.
Firstly described in patients with rheumatic fever by Sigismond Jaccoud in 1869(9), this complication has been reported in other diseases, both rheumatological (scleroderma, vasculitis, Sjögrens syndrome, psoriatic arthritis and mainly SLE) and non-rheumatological diseases (HIV infection, inflammatory bowel disease, sarcoidosis and others)(1014). This form of deforming arthritis initially resembles the ones observed in rheumatoid arthritis (RA) but, classically, it is reversible(15,16). This condition is characterized by subluxation of metacarpophalangeal joints, swan-neck and Boutonniere deformities, besides Z deformity of thumb (Figure 2). In spite of being observed principally in the hands, such deformities may involve any other joint such as knees(17), shoulders(18) and feet (19), with presence of hallux valgus, hammer toes and subluxation of metacarpophalangeal joints. There are few histological data on JA, but this condition is characterized by the presence of synovitis with inflammatory infiltrate, pericapsular fibrosis and microvascular alterations, but without the pannus classically observed in RA(20,21).
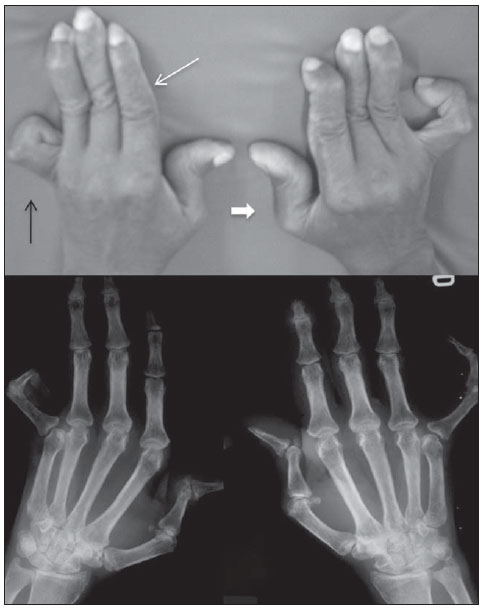
Figure 2. Jaccouds arthropathy of hands. A 55-year-old patient diagnosed with SLE for 17 years ago. A: Photo and plain radiography demonstrating findings of arthropathy such as metacarpal ulnar deviation, swan-neck deformity (fine white arrow), Boutonniere deformity (black arrow), and Z deformity of thumb (thick white arrow).
Additionally, this type of joint involvement in SLE affects the quality of life of these patients, also highlighting the absence of defined laboratory findings that may differentiate between lupus patients with and without JA(22).
Imaging findings Hands radiography demonstrates the classical deformities observed in RA, but without bone erosions. Curiously, a radiological alteration classically described in the literature would be a focal erosion on the radial aspect of the metacarpal or metatarsal head (hook erosion) determined by persistent ulnar deviation and probably representing a local adaptation to the anomalous stress produced by this bone deviation(23). However, such finding is much rarely seen in the clinical practice.
Magnetic resonance imaging (MRI) allows a more accurate visualization of the synovial and tendinous involvement that is typical of this condition. In spite of the typical presence of synovial thickening and post-contrast enhancement, the exuberant pannus caused by RA is not characterized, in general with a more subtle involvement being observed, even in the most deforming presentations of the disease. In 2003, Ostendorf et al. demonstrated by means of MRI the presence of edematous tenosynovitis and synovitis in the hands of most of their 14 lupus patients, three of them defined as JA(24). In other recent study developed in the authors institution(25), 20 patients who had met the criteria for JA of the hands were submitted to MRI and the presence of flexor tenosynovitis was demonstrated in more than 90% of the cases, as well as some degree of subarticular erosion in about 50% of the patients. This demonstrates that the absence of erosion may not be a pathognomonic finding of JA, as previously thought under the radiological point of view, and that new imaging methods can provide relevant information on this pattern of involvement not only in the assessment of hands but also of other joints (Figure 3).
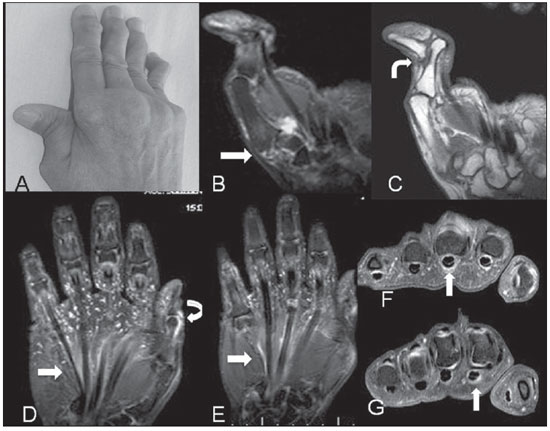
Figure 3. AC: A 60-year-old female patient diagnosed with SLE for 24 years. Photo and coronal MRI, T2-weighted image with fat suppression and T1-weighted image demonstrating Z deformity of thumb with sub-luxation and effusion in the articular interline space of the first metacarpal base trapezium (straight arrow), besides full distal interphalangeal joint luxation (curved arrow). DG: Other female patient diagnosed with SLE for 13 years. Coronal MRI T2-weighted image with fat suppression demonstrating signs of tenosynovitis (straight arrows) (D) coronal, contrastenhanced T1-weighted (E) and axial (F,G) images. Also, synovitis is observed in the interphalangeal joint of thumb (curved arrow).
Ultrasonography is a good method for recognizing and following-up synovitis, tenosynovitis and erosion, but it has been underutilized and is poorly recognized in the radiological community as a method to evaluate rheumatological diseases in general.
Rhupus hand and mild deforming arthropathy
Despite its usual non-erosive nature, some patients develop an erosive form of disease similar to RA. The term rhupus has started being employed to describe this condition because, generally, such patients meet simultaneously the criteria for classification of both SLE and RA(26). There has been a lot of discussion about to which extent ruphus hand represents a subgroup of lupus arthropathy or an association between both diseases. The possibility of overlapping between these two diseases has been estimated in 1%(27). From the imaging point of view, the condition presents the typical rheumatoid arthritis deformities with erosive involvement generally already identified at plain radiography, similarly to the findings of RA.
Mild deforming arthropathy would be a deforming, but more subtle, modality of involvement, without erosion and not meeting the criteria for classification as JA. Ulnar deviation and swan-neck deformity predominate, with no deformity at the level of the thumb and in the feet. Such sub-group has not been unanimously recognized by all the authors.
INFECTION
Infectious involvement is observed in the course of the disease, particularly in the urinary and respiratory tracts, with a prevalence > 50%, constituting one of the main causes of death and hospital admissions among these patients(28). From the musculoskeletal point of view, septic arthritis and osteomyelitis predominate, although not much frequently. Articular involvement may be observed either in a single or in multiple joints, and is primarily caused by gram-negative bacteria, particularly species of
Salmonellae and
Staphylococcus aureus, generally of hematogenic origin(29). As regards osteomyelitis, its pathogenesis in this context is also multifactorial and is related to the infectious organism virulence, to the underlying disease, to the patients immunological status as well as to the type and location of the involved bone, with
S. aureus as the main causative agent in these cases(30). So, the distribution of the infectious agents resembles the one observed in sickle-cell disease(31).
Because of the chronic use of corticoids in such patients, the signs and symptoms of infection are frequently masked and the process generally presents a chronic and indolent course. In case of persistent monoarticular arthritis, the absence of a clinical response to the therapy with corticoids or other immunosuppressive drugs should raise the suspicion of an underlying infectious process. Depending on the phase of the disease, the different imaging methods can recognize such complications and the radiologist plays a critical role in the identification of these cases (Figure 4).
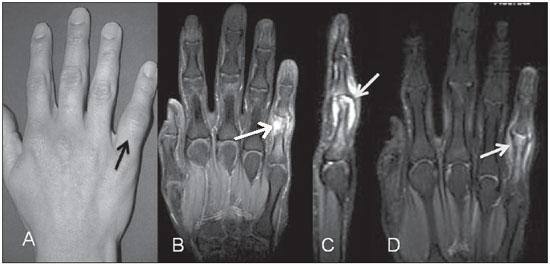
Figure 4. Septic arthritis of proximal interphalangeal joint of the fifth finger. Lupus patient with pain and focal increase in volume (A). Coronal and sagittal MRI T2-weighted images with fat suppression (B,C) demonstrating medullary edema in bone borders (black arrow on B) and strain caused by articular capsule fluid in the fifth finger proximal interphalangeal joint (white arrow on C). On the sagittal image, erosion of the corresponding proximal phalanx is also well characterized. Coronal, contrast-enhanced MRI T1-weighted image (D) demonstrating contrast enhancement surrounding the bone, synovia and soft tissues of this area.
Spontaneous tendinous rupture is a rare clinical condition. Early in the last century, McMaster demonstrated that an artificial rupture around 75% of the tendinous thickness would be unlikely to determine a full thickness rupture in the absence of a basal pathological process(32). Collagen diseases and use of corticoids, whether in association or not, would be the necessary conditions. In RA, for example, such complication is known and is particularly related to the local mechanical alteration as a result of bone erosion determining a secondary tendinous laceration.
In lupus patients, the etiology of this condition is still to be completely known and would be related to local trauma, basal chronic inflammatory process and use of corticoids(33), and no data on the actual incidence of this alteration is found in the literature. Contrary to RA, where tendinous ruptures occur almost always in the hands, in SLE such ruptures are most frequently seen in the lower limbs, affecting the quadriceps and, particularly, the patellar and Achilles tendons, also in association with a mechanical component. Corticoid therapy is known to be a predisposing factor and is present in almost all the reports in the literature. Corticoids action is related to an antimycotic effect and fibroblasts inhibition, with collagenase stimulus and consequential structural fiber disorganization(34).
Recent evidences demonstrate that this type of SLE complication may be related to a predominance of the primary inflammatory process in the tendinous sheaths leading to a focal tendinous weakness and later rupture(25). A recent systematic review developed in the authors institution has demonstrated that JA is present in at least 35% of cases of spontaneous tendinous rupture in patients with SLE (35).
OSTEONECROSIS
Osteonecrosis is a cause of morbidity and dysfunction in lupus patients with a variable incidence according to the different authors, with a prevalence ranging from 2% to 30%(36). Generally, osteonecrosis affects multiple sites, most frequently involving the femoral head (> 70% of cases) and should be always considered in cases where other areas of infarct or necrosis are identified(37). In the context of the disease, its etiology is multifactorial and still remains controversial in many cases, with several potential risk factors being considered such as presence of Raynauds phenomenon, vasculitis, hyperlipidemia and, probably, presence of antiphospholipides(38). However, high-dose corticoid therapy (> 20 mg/day) is undoubtedly the mail determining factor(39). In lupus patients, osteonecrosis determines pain previously to the joint destruction, differently from RA that occurs synchronically with joint destruction by the synovial inflammatory disease, affecting both the femoral head and the acetabular margin.
Plain radiography is generally normal in the early phase of the disease, and the presence of subchondral sclerosis already infers irreversible joint damage. Magnetic resonance imaging is the method of choice in the early diagnosis of osteonecrosis, allowing the application of therapeutic intervention(40) aimed at preventing articular collapse and the secondary degenerative disease that is the most common complication (Figure 5). Computed tomography and scintigraphy are less accurate methods and do not identify lesions in the early phase of the disease.
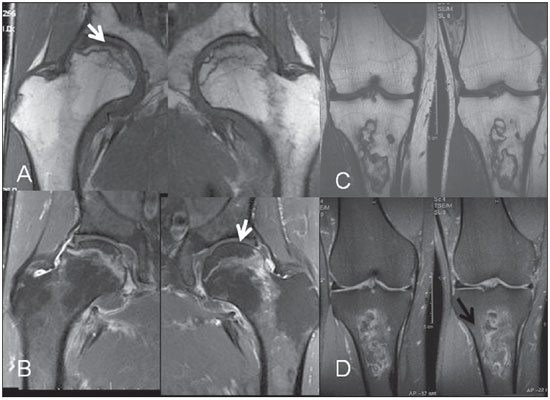
Figure 5. Radiographically occult acute avascular necrosis in a lupus patient. Coronal, right and left hip MRI T1-weighted (A) and contrast-enhanced T1-weighted image with fat suppression (B) demonstrating necrotic area with serpiginous contour (arrows) in the sub-chondral region of femoral head, with no noticeable articular surface collapse. The bilaterality of the finding is typical of association with corticoid therapy. Right knee MRI of another lupus patient under corticoid therapy, with T1-weighted (C) and T2-weighted image with fat suppression (D) demonstrating area of medullary infarct with double-line sign corresponding to the ischemic bone/reparative zone interface in the proximal metaphyseal region of the tibia (black arrow).
In 5% to 10% of lupus patients inflammatory myopathy is also observed, but laboratory findings of muscular disease may be present in up to 50% of cases(41). The pattern of inflammatory myositis associated with SLE is similar to the one of idiopathic myositis that, many times, is as severe as the primary form of the disease, making it difficult to define it as myositis secondary to SLE or as an overlapping between SLE and classical myositis(42).
Myopathy associated with use of drugs would be even more common in SLE. Additionally to corticoids which are the drugs most frequently associated with non-inflammatory myopathy, some rare cases of myopathy secondary to the use of chloroquine have been described(43). The diagnosis is generally achieved by means of clinical examination and laboratory tests, rarely requiring confirmation by muscular biopsy.
As in the cases of classical myositis, MRI is the imaging method of choice, in spite of its low specificity. This method aids in the differential diagnosis, follow-up of therapeutic response, and is useful to define the biopsy site. Typically, MRI demonstrates increased signal intensity on T2-weighted and STIR sequences as a result of the intracellular increase in the amount of fluid or inflammatory infiltrate associated with the increase in muscle volume(44). In 2000, a Brazilian study with 13 lupus patients assessed by MRI demonstrated principally predominance of muscle atrophy(45).
As in other collagen diseases, soft tissue calcifications are also identified in SLE, but they are poorly frequent in these cases(46,47). Precipitating factors like nephritis, use of alphacalcidol and even diuretic drugs have been recently related to this condition(48).
The radiological study clearly demonstrates such calcifications and, in case of dubious diagnosis, additionally to calcifications, MRI can identify an eventual association with inflammatory involvement of soft tissues.
INSUFFICIENCY FRACTURE
Many factors such as renal failure, amenorrhea, early menopause, chronic inflammatory cytokines and mainly chronic use of corticoids are involved in the genesis of osteoporosis in SLE(49). Particularly in patients with SLE, the latter is a determining factor in the development of insufficiency fractures of the spine and other sites (particularly lower limbs), with prevalence of upper osteoporotic vertebral fractures of > 20% in a recent study(50). This finding demonstrates the necessity that special attention is paid in the imaging, particularly radiological (that is the method most frequently utilized) evaluation of these patients, appreciating the findings of osteopenia and the vertebral bodies morphology. Magnetic resonance imaging is the method of choice in the early phases of the disease, when conventional radiography is not a diagnostic method yet. Classical findings of medullary bone edema are observed on the sequences with fat suppression, with linear areas of low signal intensity inside (Figure 6). Scintigraphy is also sensitive in the detection of such fractures, but it is not always very specific.
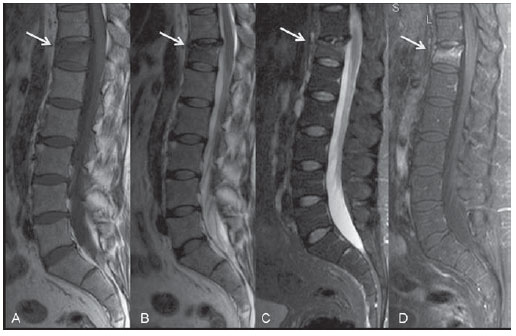
Figure 6. Acute insufficiency fracture of vertebral body. A 47-year-old lupus patient under corticoid therapy. Sagittal MRI T1-weighted (A) and FSE T2-weighted (B) images demonstrate partial T12 upper plateau collapse (arrows). The edema becomes better characterized on the STIR sequence (C), associated with a local reactional inflammatory process with contrast uptake on the T1-weighted image (D).
Musculoskeletal involvement is frequently observed in lupus disease and may be an early indicator of disease activity. The degree of involvement may range from a subtle arthralgia to marked presentations of deforming arthropathy, besides tendinous ruptures and other forms of involvement such as myopathy, osteonecrosis, insufficiency fractures and infection, the latter being most frequently associated with the adopted treatment.
Although deforming arthritis (JA) is classically reversible, it is important to highlight its relevance in the musculoskeletal involvement of SLE, as it may be largely confused with the clinical picture of RA. In this context, the radiologist plays an extremely relevant role, calling the attention of the clinician to this hypothesis represented by plain radiographic findings corresponding to significant deformities and subluxation and the virtual absence of erosions.
Another significant contribution of the radiological evaluation of the musculoskeletal system in patients with SLE was achieved with a more frequent utilization of MRI so that the development of avascular necrosis secondary to prolonged corticoid therapy is early detected, allowing the adoption of measures aimed at avoiding bone collapse.
Other imaging method that has gained considerable ground in the follow-up of rheumatological patients, particularly those with RA, is high-definition ultrasonography of joints. The experience of this method in SLE patients is extremely limited. A study is currently under development in the authors institution aiming at evaluating the relevance of ultrasonography for patients with JA, besides determining the distribution and severity of alterations in these patients as compared with lupus patients with arthritis, but without JA.
Thus, with a deeper knowledge on radiological findings in patients with SLE, the radiologist takes over a differentiated role in the follow-up of these patients as he/she already does in cases of rheumatological conditions such as RA.
REFERENCES
1. Lebowitz R, Schumacher HR Jr. Articular manifestations of systemic lupus erythematosus. Ann Intern Med. 1971;74:91121.
2. Tan EM, Cohen AS, Fries JF, et al. The 1982 revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1982;25:12717.
3. Dubois EL, Tuffanelli DL. Clinical manifestations of systemic lupus erythematosus. JAMA. 1964;190:10411.
4. Reilly PA, Evison G, McHugh NJ, et al. Arthropathy of hands and feet in systemic lupus erythematosus. J Rheumatol. 1990;17:77784.
5. Aptekar RG, Lawless OJ, Decker JL. Deforming non-erosive arthritis of the hand in systemic lupus erythematosus. Clin Orthop Relat Res. 1974;(100):1204.
6. Sierra-Jimenez G, Sanchez-Ortiz A, Aceves-Avila FJ, et al. Tendinous and ligamentous derangements in systemic lupus erythematosus. J Rheumatol. 2008;35:218791.
7. Bywaters EGL. Jaccouds syndrome: a sequel to the joint involvement of systemic lupus erythematosus. Clin Rheum Dis. 1975;1:12548.
8. van Vugt RM, Derksen RH, Kater L, et al. Deforming arthropathy or lupus and rhupus hands in systemic lupus erythematosus. Ann Rheum Dis. 1998;57:5404.
9. Manthorpe R. The man behind the syndrome. Sigismond Jaccoud. With his 23d lecture he became part of medical history. Lakartidningen. 1992;89:15856.
10. Amano H, Furuhata N, Tamura N, et al. Hypocomplementemic urticarial vasculitis with Jaccouds arthropathy and valvular heart disease: case report and review of the literature. Lupus. 2008;17:83741.
11. Ballard M, Meyer O, Adle-Biassette H, et al. Jaccouds arthropathy with vasculitis and primary Sjögrens syndrome. A new entity. Clin Exp Rheumatol. 2006;24(2 Suppl 41):S1023.
12. Ben Miled-MRad K, MRad S, Kchir M, et al. Carolis disease and Jaccouds arthropathy. Ann Gastroenterol Hepatol (Paris). 1993;29:1079.
13. Bradley JD, Pinals RS. Jaccouds arthropathy in scleroderma. Clin Exp Rheumatol. 1984;2:33740.
14. Conrozier T, Balblanc JC, Chapard R, et al. Jaccouds arthritis and angioimmunoblastic lymphadenopathy. Rev Rhum Mal Osteoartic. 1990;57:4235.
15. Santiago MB, Galvão V. Jaccoud arthropathy in systemic lupus erythematosus: analysis of clinical characteristics and review of the literature. Medicine (Baltimore). 2008;87:3744.
16. Caznoch CJ, Esmanhotto L, Silva MB, et al. Padrão de comprometimento articular em pacientes com lúpus eritematoso sistêmico e sua associação com presença de fator reumatóide e hiperelasticidade. Rev Bras Reumatol. 2006;46:2615.
17. De la Sota M, Maldonado Cocco JA. Jaccouds arthropathy in knees in systemic lupus erythematosus. Clin Rheumatol. 1989;8:4167.
18. Siam AR, Hammoudeh M. Jaccouds arthropathy of the shoulders in systemic lupus erythematosus. J Rheumatol. 1992;19:9801.
19. Morley KD, Leung A, Rynes RI. Lupus foot. Br Med J (Clin Res Ed). 1982;284:5578.
20. Spronk PE, ter Borg EJ, Kallenberg CG. Patients with systemic lupus erythematosus and Jaccouds arthropathy: a clinical subset with an increased C reactive protein response? Ann Rheum Dis. 1992;51:35861.
21. Paredes JG, Lazaro MA, Citera G, et al. Jaccouds arthropathy of the hands in overlap syndrome. Clin Rheumatol. 1997;16:659.
22. Galvão V, Atta AM, Sousa Atta ML, et al. Profile of autoantibodies in Jaccouds arthropathy. Joint Bone Spine. 2009;76:35660.
23. Pastershank SP, Resnick D. Hook erosions in Jaccouds arthropathy. J Can Assoc Radiol. 1980;31:1745.
24. Ostendorf B, Scherer A, Specker C, et al. Jaccouds arthropathy in systemic lupus erythematosus: differentiation of deforming and erosive patterns by magnetic resonance imaging. Arthritis Rheum. 2003;48:15765.
25. Ribeiro DS, Galvão V, Fernandes JL, et al. Magnetic resonance imaging of Jaccouds arthropathy in systemic lupus erythematosus. Joint Bone Spine. 2010;77:2415.
26. Fernández A, Quintana G, Rondón F, et al. Lupus arthropathy: a case series of patients with rhupus. Clin Rheumatol. 2006;25:1647.
27. Mawson AR. Are rheumatoid arthritis and systemic lupus erythematosus inversely related diseases? Med Hypotheses. 1985;18:37786.
28. Petri M. Infection in systemic lupus erythematosus. Rheum Dis Clin North Am. 1998;24:42356.
29. Chen JY, Luo SF, Wu YJ, et al. Salmonella septic arthritis in systemic lupus erythematosus and other systemic diseases. Clin Rheumatol. 1998;17:2827.
30. Cuchacovich R, Gedalia A. Pathophysiology and clinical spectrum of infections in systemic lupus erythematosus. Rheum Dis Clin North Am. 2009;35:7593.
31. Epss CH, Bryant DD, Coles MJ, et al. Osteomyelitis in patients who have sickle-cell disease. Diagnosis and management. J Bone Joint Surg Am. 1991;73:128194.
32. McMaster PE. Tendon and muscle rupture. Clinical and experimental studies on the causes and location of subcutaneous ruptures. J Bone Joint Surg. 1933;15:70522.
33. Kissel CG, Sundareson AS, Unroe BJ. Spontaneous Achilles tendon rupture in a patient with systemic lupus erythematosus. J Foot Surg. 1991;30:3907.
34. Houck JC, Patel YM. Proposed mode of action of corticosteroids on the connective tissue. Nature. 1965;206:15860.
35. Alves EM, Macieira JC, Borba E, et al. Spontaneous tendon rupture in systemic lupus erythematosus: association with Jaccouds arthropathy. Lupus. 2010;19:24754.
36. Mont MA, Glueck CJ, Pacheco IH, et al. Risk factors for osteonecrosis in systemic lupus erythematosus. J Rheumatol. 1997;24:65462.
37. Cozen L, Wallace DJ. Avascular necrosis in systemic lupus erythematosus: clinical associations and a 47-year perspective. Am J Orthop (Belle Mead NJ). 1998;27:3524.
38. Campos LM, Kiss MH, DAmico EA, et al. Antiphospholipid antibodies and antiphospholipid syndrome in 57 children and adolescents with systemic lupus erythematosus. Lupus. 2003;12:8206.
39. Zizic TM, Marcoux C, Hungerford DS, et al. Corticosteroid therapy associated with ischemic necrosis of bone in systemic lupus erythematosus. Am J Med. 1985;79:596604.
40. Alves EM, Angrisani AT, Santiago MB. The use of extracorporeal shock waves in the treatment of osteonecrosis of the femoral head: a systematic review. Clin Rheumatol. 2009;28:124751.
41. Isenberg D. Myositis in other connective tissue disorders. Clin Rheum Dis. 1984;10:15174.
42. Garton MJ, Isenberg DA. Clinical features of lupus myositis versus idiopathic myositis: a review of 30 cases. Br J Rheumatol. 1997;36:106774.
43. Richter JG, Becker A, Ostendorf B, et al. Differential diagnosis of high serum creatine kinase levels in systemic lupus erythematosus. Rheumatol Int. 2003;23:31923.
44. Adams EM, Chow CK, PremkumarA, et al. The idiopathic inflammatory myopathies: spectrum of MR imaging findings. Radiographics. 1995;15:56374.
45. Hilário MO, Yamashita H, Lutti D, et al. Juvenile idiopathic inflammatory myopathies: the value of magnetic resonance imaging in the detection of muscle involvement. Sao Paulo Med J. 2000;118:3540.
46. Souza RAS, Rangel LV, Souza HFS, et al. Lúpus eritematoso cutâneo e calcinose universalis. Rev Bras Reumatol. 2000;40:1820.
47. Souza HFS, Souza RAS, Rangel LV, et al. Calcificação intracerebral em lúpus eritematoso sistêmico. Rev Bras Reumatol. 2001;41:1236.
48. Okada J, Nomura M, Shirataka M, et al. Prevalence of soft tissue calcifications in patients with SLE and effects of alfacarcidol. Lupus. 1999;8:45661.
49. Panopalis P, Yazdany J. Bone health in systemic lupus erythematosus. Curr Rheumatol Rep. 2009;11:17784.
50. Bultink IE, Lems WF, Kostense PJ, et al. Prevalence of and risk factors for low bone mineral density and vertebral fractures in patients with systemic lupus erythematosus. Arthritis Rheum. 2005;52:204450.
1. Specialist in Imaging Diagnosis, MD, Radiologist at Clínica Image Memorial and at Hospital Santa Izabel, PhD Fellow degree at Escola Bahiana de Medicina e Saúde Pública, Salvador, BA, Brazil.
2. Professor of Radiology, Faculdade de Medicina da Bahia/UFBA, Clinical Director, Clínica Image Memorial, Salvador, BA, Brazil.
3. Specialist in Imaging Diagnosis, Clinical Director, Clínica Image Memorial, Salvador, BA, Brazil.
4. Master, Physiotherapist, Professor at Universidade Católica do Salvador, Salvador, BA, Brazil.
5. PhD, Associate Professor, Escola Bahiana de Medicina e Saúde Pública, Coordinator for the Service of Rheumatology of Hospital Santa Izabel, Salvador, BA, Brazil.
Mailing Address:
Dr. Daniel Sá Ribeiro
Rua Pacífico Pereira, 590/101, Garcia
Salvador, BA, Brazil, 40100-170
E-mail: danielribeiro.sa@gmail.com
Received April 18, 2010.
Accepted after revision September 13, 2010.
 Vol. 44 nº 1 - Jan. /Feb. of 2011
Vol. 44 nº 1 - Jan. /Feb. of 2011






